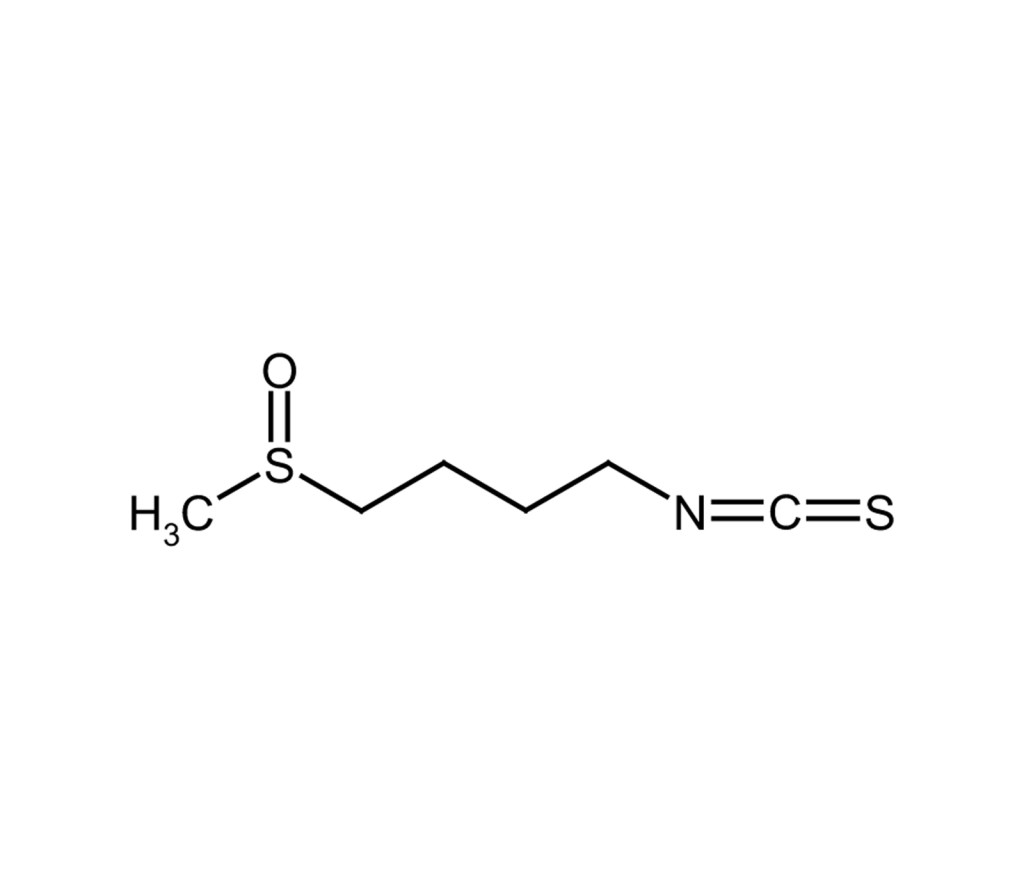
Carcinogenesis inhibitor
R,S-Sulforaphane is a synthetic version of the naturally occurring compound, R-sulforaphane, from Broccoli. It is a racemic mixture of both the R and S isomers.
Potent, selective inducer of phase II detoxification enzymes. Inhibits chemically induced mammary tumor formation in rats. Inhibits LPS-induced HMGB1 (high mobility group box 1) secretion.
Shipping: Available products typically ship within 24/48h, via priority shipping.
Do you need support? Contact Customer Service or Technical Support.
Online Account
Access or Create Your Account
Product Details
| Alternative Name |
D,L-Sulforaphane, 1-Isothiocyanato-4-methylsulfinyl-butane |
|---|---|
| Appearance |
Slightly yellowish liquid. |
| CAS |
4478-93-7 (unspecified stereo) |
| Formula |
C6H11NOS2 |
| Identity |
Identity determined by 1H-NMR and MS. |
| MI |
14: 8962 |
| MW |
177.3 |
| Purity |
≥98% (UHPLC) |
| Solubility |
Soluble in methanol or DMSO. |
| Source |
Synthetic. |
Handling & Storage
| Use/Stability |
As indicated on product label or CoA when stored as recommended. |
|---|---|
| Long Term Storage |
-20°C |
| Shipping |
Ambient Temperature |
| Regulatory Status |
RUO – Research Use Only |
|---|
- Dietary Antioxidants Significantly Attenuate Hyperoxia-Induced Acute Inflammatory Lung Injury by Enhancing Macrophage Function via Reducing the Accumulation of Airway HMGB1: Patel, V., Dial, K., et al.; Int. J. Mol. Sci. 21, (2020), Abstract
- Free light chains injure proximal tubule cells through the STAT1/HMGB1/TLR axis: Upadhyay, R., Ying, W. Z., et al.; JCI Insight 5, (2020), Abstract
- Simultaneous quantification of sulforaphene and sulforaphane by reverse phase HPLC and their content in Raphanus sativus L. var. caudatus Alef extracts: S. Sangthong, et al.; Food. Chem. 201, 139 (2016), Application(s): Standard, Abstract
- Flavokawains A and B from kava (Piper methysticum) activate heat shock and antioxidant responses and protect against hydrogen peroxide-induced cell death in HepG2 hepatocytes: Pinner, K. D., Wales, C. T., et al.; Pharm. Biol. 54, 1503 (2016), Abstract
- Synergy between sulforaphane and selenium in protection against oxidative damage in colonic CCD841 cells: Y. Wang, et al.; Nutr. Res. 35, 610 (2015), Abstract
- 4-Methylthiobutyl isothiocyanate (Erucin) from rocket plant dichotomously affects the activity of human immunocompetent cells: C. Gründemann, et al.; Phytomedicine 22, 369 (2015), Application(s): Gas Chromatography – Mass Spectrometry , Abstract
- Protective effect of sulforaphane against oxidative stress: Recent advances: C.E. Guerrero-Beltran, et al.; Exp. Toxicol. Pathol. 64, 503 (2012), Abstract
- Sulforaphane targets pancreatic tumor-initiating cells by NF-{kappa}B-induced anti-apoptotic signaling: G. Kallifatidis, et al.; Gut 58, 949 (2009), Abstract
- The phase 2 enzyme inducers ethacrynic acid, DL-sulforaphane, and oltipraz inhibit lipopolysaccharide-induced high-mobility group box 1 secretion by RAW 264.7 cells: M.E. Killeen, et al.; J. Pharmacol. Exp. Ther. 316, 1070 (2006), Abstract
- A novel mechanism of chemoprotection by sulforaphane: inhibition of histone deacetylase: M.C. Myzak, et al.; Cancer Res. 64, 5767 (2004), Abstract — Full Text
- Anticarcinogenic activities of sulforaphane and structurally related synthetic norbornyl isothiocyanates: Y. Zhang, et al.; PNAS 91, 3147 (1994), Abstract — Full Text
- A major inducer of anticarcinogenic protective enzymes from broccoli: isolation and elucidation of structure: Y. Zhang, et al.; PNAS 89, 2399 (1992), Abstract — Full Text
Related Products
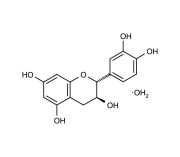
| Alternative Name | (+)-3,3’,4’,5,7-Flavanpentol . H2O |
|---|---|
| CAS | 225937-10-0 |
| Purity | ≥98% (HPLC) |
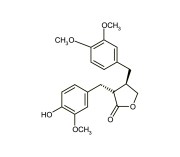
| Alternative Name | (3R,4R)-4-[(3,4-Dimethoxyphenyl)methyl]dihydro-3-[(4-hydroxy-3-methoxyphenyl)methyl]-2-(3H)-furanone |
|---|---|
| CAS | 7770-78-7 |
| Couple Type | Inhibitor |
| Purity | ≥97% (HPLC) |
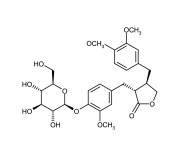
| Alternative Name | (3R,4R)-4-[(3,4-Dimethoxyphenyl)methyl]-3-[[4-(β-D-glucopyranosyloxy)-3-methoxyphenyl]methyl]dihydro-2(3H)-furanone |
|---|---|
| CAS | 20362-31-6 |
| Couple Type | Inhibitor |
| Purity | ≥95% (HPLC) |
| Alternative Name | EGCG, (2R,3R)-2-(3,4,5-Trihydroxyphenyl)-3,4-dihydro-1[2H]-benzopyran-3,5,7-triol 3-(3,4,5-trihydroxybenzoate) |
|---|---|
| CAS | 989-51-5 |
| Couple Type | Inhibitor |
| Purity | ≥98% (HPLC) |
Last modified: May 29, 2024
Datasheet, Manuals, SDS & CofA
Manuals And Inserts
Certificate of Analysis
Please enter the lot number as featured on the product label
SDS
Enzo Life Science provides GHS Compliant SDS
If your language is not available please fill out the SDS request form
 Lab Essentials
Lab Essentials AMPIVIEW® RNA probes
AMPIVIEW® RNA probes Enabling Your Projects
Enabling Your Projects  GMP Services
GMP Services Bulk Solutions
Bulk Solutions Research Travel Grant
Research Travel Grant Have You Published Using an Enzo Product?
Have You Published Using an Enzo Product?