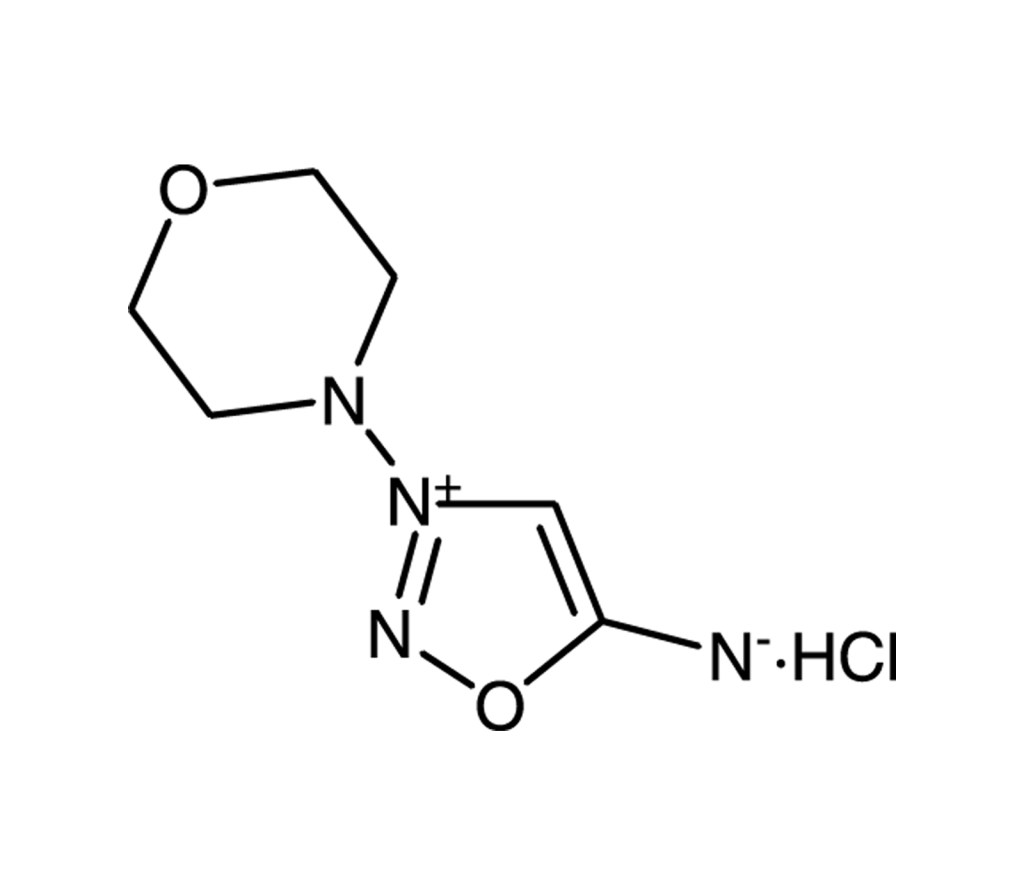
Releases NO and superoxide anion under physiological conditions
SIN-1 is a metabolite of molsidomine which decomposes spontaneously in solution in the presence of oxygen, releasing NO and superoxide anion under physiological conditions. Since NO and superoxide react to form peroxynitrite, SIN-1 may actually produce peroxynitrite under physiological conditions. It is a potent vasodilator and inhibitor of platelet aggregation and produces significant protective effects in myocardial ischemia-reperfusion. The effects of SIN-1 are markedly attenuated by nitroglycerin-induced tolerance. Inhibits cysteine proteases.
Shipping: Available products typically ship within 24/48h, via priority shipping.
Do you need support? Contact Customer Service or Technical Support.
Online Account
Access or Create Your Account
Product Details
| Alternative Name |
SIN-1, Linsidomine |
|---|---|
| Appearance |
White to off-white solid. |
| CAS |
33876-97-0 |
| Couple Target |
Protease |
| Couple Type |
Inhibitor |
| Formula |
C6H10N4O2 . HCl |
| Identity |
Determined by NMR. |
| MW |
206.6 |
| MeltingPoint |
186-187°C |
| Purity |
≥98% (HPLC) |
| Solubility |
Soluble in water (25mg/ml), 100% ethanol or DMSO (>5mg/ml). |
Handling & Storage
| Use/Stability |
As indicated on product label or CoA when stored as recommended. |
|---|---|
| Handling |
Use freshly prepared solutions. |
| Long Term Storage |
-20°C |
| Shipping |
Ambient Temperature |
| Regulatory Status |
RUO – Research Use Only |
|---|
- (2-Aminobenzothiazole)-Methyl-1,1-Bisphosphonic Acids: Targeting Matrix Metalloproteinase 13 Inhibition to the Bone: A. Laghezza, et al.; Pharmaceuticals (Basel) 14, 85 (2021), Abstract
- Tissue-specific contributions of Tmem79 to atopic dermatitis and mast cell-mediated histaminergic itch: J.J. Emrick, et al.; PNAS 115, 12091 (2018), Application(s): Neuronal cell culture, Abstract — Full Text
- Modification of HSP proteins and Ca2+ are responsible for the NO-derived peroxynitrite mediated neurological damage in PC12 cell: J. Wen, et al.; Int. J. Clin. Exp. Pathol. 8, 4492 (2015), Abstract — Full Text
- EPR detection of cellular and mitochondrial superoxide using cyclic hydroxylamines: S.I. Dikalov, et al.; Free Rad. Res. 45, 417 (2011), Abstract
- Interactions of peroxynitrite with uric acid in the presence of ascorbate and thiols: implications for uncoupling endothelial nitric oxide synthase: N. Kuzkaya et al.; Biochem. Pharmacol. 70, 343 (2005), Abstract
- New glycosidase activated nitric oxide donors: glycose and 3-morphorlinosydnonimine conjugates: T.B. Cai et al.; J. Org. Chem. 70, 3518 (2005), Abstract
- In vitro cytotoxicity of glyco-S-nitrosothiols. a novel class of nitric oxide donors: H. Babich & H.L. Zuckerbaun; Toxicol. In Vitro 15, 181 (2001), Abstract
- An activity in rat tissues that modifies nitrotyrosine-containing proteins: Y. Kamisaki et al.; PNAS 95, 11584 (1998), Abstract
- Nitric oxide regulates cardiac Ca2+ current. Involvement of cGMP-inhibited and cGMP-stimulated phosphodiesterases through guanylyl cyclase activation: P.F. Mery et al.; J. Biol. Chem. 268, 26286 (1993), Abstract
- Neuronal growth cone collapse and inhibition of protein fatty acylation by nitric oxide: D.T. Hess et al.; Nature 366, 562 (1993), Abstract
- A redox-based mechanism for the neuroprotective and neurodestructive effects of nitric oxide and related nitroso-compounds: S.A. Lipton et al.; Nature 364, 626 (1993), Abstract
- Effect of nitric oxide production on the redox modulatory site of the NMDA receptor-channel complex: S.Z. Lei et al.; Neuron 8, 1087 (1992), Abstract
- Cardioprotection and attenuation of endothelial dysfunction by organic nitric oxide donors in myocardial ischemia-reperfusion: M.R. Siegfried et al.; J. Pharmacol. Exp. Ther. 260, 668 (1992), Abstract
- Production of hydroxyl radicals from the simultaneous generation of superoxide and nitric oxide: N. Hogg et al.; Biochem. J. 281, 419 (1992), Abstract
- Nitroglycerin-induced tolerance affects multiple sites in the organic nitrate bioconversion cascade: P.J. Henry et al.; J. Pharmacol. Exp. Ther. 248, 762 (1989), Abstract
- Molsidomine: alternative approaches to treat myocardial ischemia: R.-E. Nitz & V.B. Fiedler; Pharmacotherapy 7, 28 (1987), Abstract
- Regulation of cyclic GMP formation by soluble guanylate cyclase: stimulation by NO-containing compounds: E. Bohme et al.; Adv. Cyclic Nucleotide Protein Phosphorylation Res. 17, 259 (1984), Abstract
- Inhibition of platelet aggregation and stimulation of guanylate cyclase by an antianginal agent molsidomine and its metabolites: M. Nishikawa et al.; J. Phar.macol. Exp. Ther. 220, 183 (1982), Abstract
Related Products
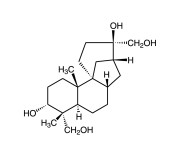
| CAS | 38966-21-1 |
|---|---|
| Couple Type | Inhibitor |
| Purity | ≥98% (TLC) |
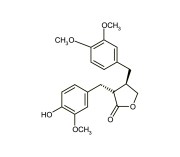
| Alternative Name | (3R,4R)-4-[(3,4-Dimethoxyphenyl)methyl]dihydro-3-[(4-hydroxy-3-methoxyphenyl)methyl]-2-(3H)-furanone |
|---|---|
| CAS | 7770-78-7 |
| Couple Type | Inhibitor |
| Purity | ≥97% (HPLC) |
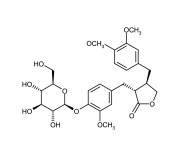
| Alternative Name | (3R,4R)-4-[(3,4-Dimethoxyphenyl)methyl]-3-[[4-(β-D-glucopyranosyloxy)-3-methoxyphenyl]methyl]dihydro-2(3H)-furanone |
|---|---|
| CAS | 20362-31-6 |
| Couple Type | Inhibitor |
| Purity | ≥95% (HPLC) |
| Alternative Name | (3α,16α)-Eburnamenin-14(15H)-one, Vincamone, Vinburnine |
|---|---|
| CAS | 4880-88-0 |
| Purity | ≥99% (Assay) |
Last modified: May 29, 2024
Datasheet, Manuals, SDS & CofA
Manuals And Inserts
Certificate of Analysis
Please enter the lot number as featured on the product label
SDS
Enzo Life Science provides GHS Compliant SDS
If your language is not available please fill out the SDS request form
 Lab Essentials
Lab Essentials AMPIVIEW® RNA probes
AMPIVIEW® RNA probes Enabling Your Projects
Enabling Your Projects  GMP Services
GMP Services Bulk Solutions
Bulk Solutions Research Travel Grant
Research Travel Grant Have You Published Using an Enzo Product?
Have You Published Using an Enzo Product?